ez-SourceDocx
ez-SourceDocx transforms clinical trials by enabling seamless eSource and eCRF data capture through a user-friendly tablet app. Facilitating direct data input during patient visits, it enhances data integrity with built-in validations, minimizes study costs and risks, and streamlines workflows, ensuring efficient protocol execution and optimal site performance.
Top ez-SourceDocx Alternatives

StackScan
Unlock deep insights into website technologies with StackScan, tracking 50,000+ tools (450+ technology categories to explore).
Entrypoint i4
Entrypoint i4 empowers users to effortlessly design, deploy, and manage custom data entry applications.
Korio
Korio serves as a cutting-edge Randomization & Trial Supply Management (RTSM) platform, expertly designed to adapt to the complexities of modern clinical trials.
eCTMS
eCTMS is an advanced Clinical Trial Management Software designed to streamline trial processes, enhance data accuracy, and facilitate real-time collaboration among research teams.
Labfront
Labfront revolutionizes clinical trial management by seamlessly aggregating physiological data from wearables, survey responses, and adherence records.
DFdiscover
DFdiscover is an agile clinical trial management software that enhances data collection from diverse sources, including EDC and paper.
Leapcure
Leapcure, a pioneering patient recruitment and engagement platform, enhances clinical trial participation by connecting sponsors with eligible patients.
Datacubed Health
Its adaptable application ensures participants easily understand protocols while maintaining active involvement through a proprietary...
Lokavant
The platform offers real-time scenario analysis, empowering teams to navigate complexities while optimizing trial performance...
Clinical StudyPal
It facilitates efficient patient recruitment, real-time communication, and comprehensive data collection through user-friendly apps and...
Clinical Concepts
Their modular solutions streamline patient recruitment, real-time data collection, and subject randomization while providing robust...
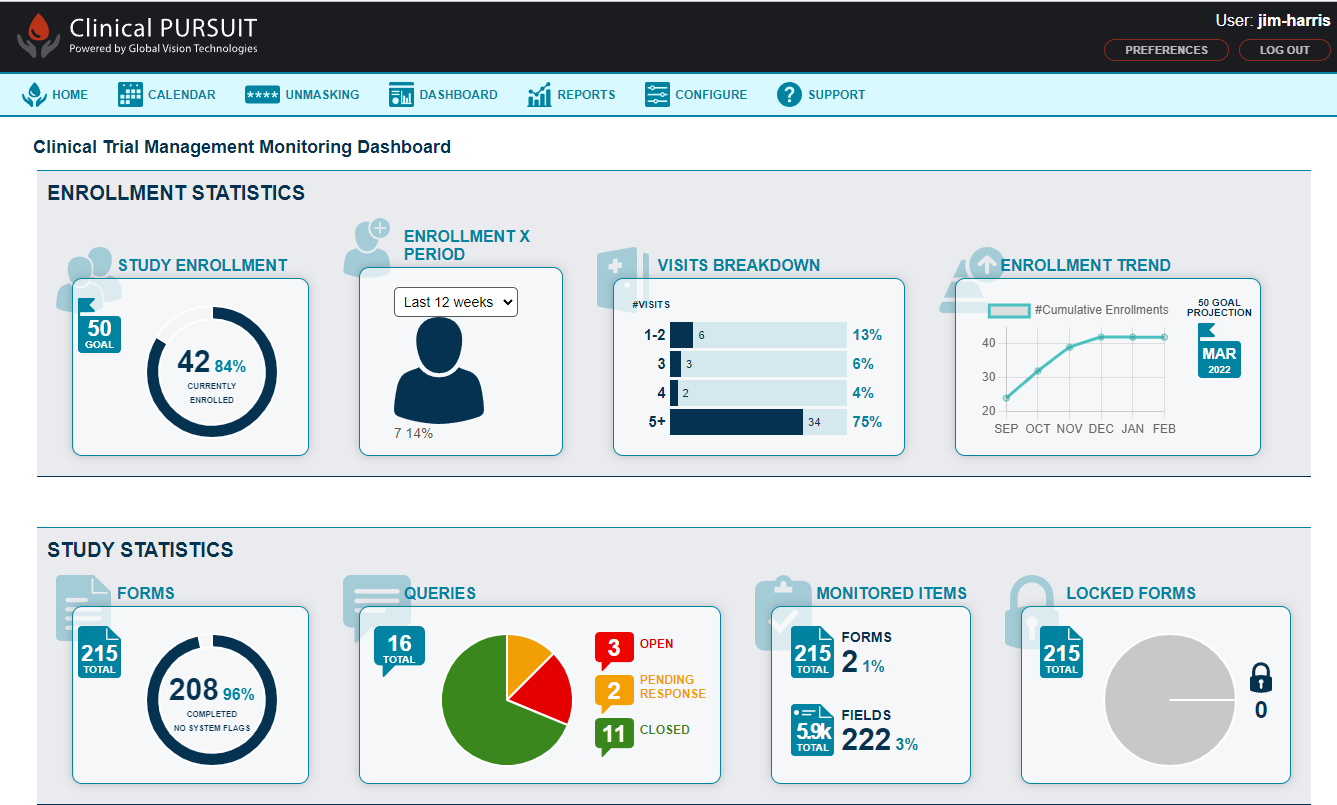
ClinicalPURSUIT
By utilizing a web-based solution, it significantly reduces study costs while ensuring secure data management...
PRA Prism
Its zero footprint design allows users to access the platform effortlessly on any device, ensuring...
Jeeva eClinical Cloud
It supports real-time data collection, remote patient engagement, and efficient scheduling, allowing researchers to adjust...
Protocol Manager (CTMS)
By implementing standardized operating procedures, it ensures compliance with FDA’s 21 CFR part 11 regulations...
SimpleTrials
It integrates CTMS and eTMF functionalities into a single platform, catering to specialized studies and...
Top ez-SourceDocx Features
- Tablet-based data capture
- Integration with external EDC systems
- Built-in edit and validation checks
- Real-time remote monitoring
- Streamlined clinical workflows
- Protocol execution guidance
- High-quality data collection
- Reduced data entry errors
- Fast study completion
- Cost-effective trial management
- User-friendly interface
- Enhanced investigator evaluation
- Reduced study risks
- Automated data validation
- Multi-location accessibility
- Customizable data capture forms
- Patient visit data synchronization
- Improved site efficiency
- FDA-endorsed processes
- Comprehensive training and support